Introduction
Click for full PDF publication

- Intro
- Pest Management Principles
- Sampling Soybeans to Estimate Insect Densities and Plant Damage
- Visual Inspection Sampling Method
- Maturity Groups, Determinate and Indeterminate Soybeans, and Growth Stages
Authors:
Suhas S. Vyavhare | Postdoctoral Research Associate
Michael O. Way | Professor of Entomology
Allen Knutson | Professor and Extension Entomologist
Stephen Biles | Extension Agent-Integrated Pest Management
Rebecca A. Pearson | Research Assistant
Texas A&M University System
Intro
Managing soybean insect pests in Texas depends largely on geography, climate, and common agronomic practices.
Most soybeans in Texas are grown in the Northern High Plains, Blacklands, Northeast Texas, Upper Gulf Coast, and the Lower Rio Grande Valley—an area that extends north to south from Oklahoma to the Mexican border and west to east from the Northern High Plains to the Louisiana border.
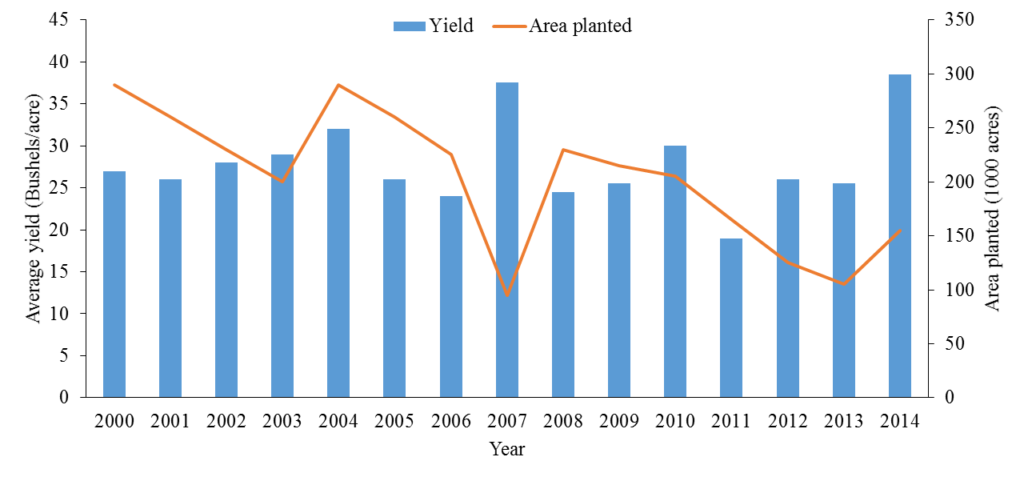
Research data show that, over the last decade, there have been substantial fluctuations in both the area planted and yields produced (Fig. 1).
Market prices and climate often influence both acreage and yield. Based on a 2012 census, less than 20 percent of Texas soybean acre- age is irrigated, with most soybeans produced in areas with adequate rainfall (generally in the eastern half of the state where annual precipitation can exceed 40 inches). Recently, soybean production has increased in the Lower Rio Grande Valley where two irrigated crops are produced each year. Soybeans in the Northern High Plains are also irrigated and frequently planted following an early spring failure of the co on crop. e average yield of irrigated soybeans in Texas is almost twice that of rain-fed soybeans.
But, soybeans do not tolerate excessively wet conditions such as standing water. Heavy, persistent rains that result in prolonged saturated soil can kill soybeans or cause severe stunting. A good management option is to plant soybeans on beds rather than drill-planting, particularly on the Upper Gulf Coast where rainfall is relatively abundant. Also, precision-leveled fields can improve drainage and reduce irrigation costs.
Soybean farmers should manage their crop to achieve maximum yield at minimum cost. is requires a high level of decision-making, similar to more high-value crops such as rice and co on. Management decisions involve variety selection, planting method and date, and pest control programs. Weed, disease, and insect control programs must be effective and timely, which means the soybean farmer or crop consultant must do the necessary agronomic “homework” and scout fields regularly and thoroughly from planting to harvest.

Soybeans are grown in diverse regions of Texas and the complex of insects across the state that a back soybeans is also diverse. For each region, a unique group of insect pests damages soybeans, making the crop vulnerable to attack throughout the crop season. is is why frequent and careful scouting for insect pests and bene cial insects is critical to successful soybean production (Fig. 2).
High spring and summer temperatures and humidity, abundant rainfall, and a long growing season (characteristic of the eastern half of the state) are conducive to rapid buildup of pest populations. A soybean farmer who does not check fields frequently runs the risk of being unaware of damaging pest populations during critical growth stages. is lack of attention and management can result in significant yield and quality loss and less than optimum insect control.
Unfortunately, some farmers apply insecticides on a “calendar basis,” causing an overuse of pesticides and detrimental effects on the environment and beneficial species. Also, applying insecticides without scouting can needlessly increase production costs and foster resistance development.
Pest Management Principles
Integrated pest management (IPM) is a philosophy used in the design of insect, mite, disease, and weed control programs. It uses the most economically and ecologically sound combination of e ective pest suppression techniques to create an economically viable production management system.
e rst line of defense against pests is prevention through good agronomic practices or cultural methods that discourage pest population development. Implement control measures only when pest populations reach levels that cause crop losses greater than the cost of the treatment. is is the economic threshold level or treatment threshold. Scout elds regularly to determine if and when that level is reached. Precise timing and execution of each production operation is essential. In short, IPM strives to optimize rather than maximize pest control efforts.
e treatment thresholds in this publication are guidelines. Several factors a ect the level of damage soybean plants can tolerate before the cost of a control tactic (such as the use of an insecticide) becomes pro table. These factors include the anticipated market value of the crop, anticipated yield, and the cost of the treatment.
In general, when the market value of soybeans is high and the cost of control is low, treatment thresholds decrease (fewer pests or less
Sampling Soybeans to Estimate Insect Densities and Plant Damage
In order to know when pest infestations exceed the economic or treatment threshold, estimate the densities of insect pests and damage in a field. Sampling thoroughly at least once a week is the key to effec- tive pest management (Fig. 3).

Do not bias results by sampling only near the field margins. Often, pest populations will be greater close to the edge of a field. It takes more effort to sample deeper into the field, but the results are more accurate and representative of the entire field.
The more samples you take, the more accurate the estimate of target pest densities will be. Do not sample only one area of a field; take samples from all portions of it. For instance, if a soybean field is rectangular, the minimum sampling effort should involve all four sides of the field.
Obvious field differences such as soil type, surrounding vegetation patterns, and topography can impact insect populations. The more variable the field, the more samples you will need to take to account for these differences. Also, sample more areas if the results show that pest densities or damage are close to treat- ment thresholds or if the field is very large.
There are various ways to sample and, based on the production practices in a given field, each method has its strengths and weaknesses. Regardless of the sampling method, scout soybeans frequently and thoroughly from planting to near maturity because pest densities can rapidly increase to damaging levels. A Texas soybean farmer once quipped, “The best thing you can put on your field is your shadow!”
Visual Inspection Sampling Method
You can learn a lot by simply observing insects and damage in soybean fields. Many farmers routinely inspect fields visually without using a sampling tool. For instance, after seedlings emerge, main stem girdling by threecornered alfalfa hopper can cause young plants to fall over, reducing plant stand. This type of damage is easy to spot and should alert the scout (farmer or crop consultant) to perform a more detailed assessment throughout the field.
The larvae of the lesser cornstalk borer feeds below or at the surface of the soil and can kill seedling plants. Groups of dead plants within the same or adjacent rows warrants further inspection for lesser cornstalk borers.
Increased leaf damage over a relatively short time can indicate expanding densities of velvetbean caterpillars, green cloverworms, and soybean loopers or cabbage loopers. In the late summer, these caterpillars infest soybeans grown in the eastern half of the state. If scouts inspect a field at least once a week, they will notice an increase in leaf damage and conduct more intensive sampling.
Sweep Net Sampling Method
Sweep net sampling is quick, covers a relatively large area, and is easy to use on soybeans planted on beds or drill-planted (Fig. 4).
For sweep net sampling:
- Use a 15-inch diameter sweep net attached to a 2-foot wooden
- Pass the sweep net through the soybean foliage so that the top of the sweep net is even with the top of the soybean plant
- Draw the net through the foliage in 180-degree sweeps while walking through the
- With each step, swing the net from side to side across the A sample should consist of at least 10 consecutive sweeps.
- Shake the contents of the sweep net down to the bottom of the
- Count the captured insects as you slowly open the net or place them in plastic bags for later identification and countin
- Repeat the sampling procedure in at least 10 random
Advantages:
- Sweep nets are easy to use, relatively inexpensive, and easy to carry into and out of the
- Fewer specimens are lost compared to using a ground cloth.

Disadvantages:
- This method does not sample the bottom portions of the soybeans (below the bottom of the net).
- Seedling and small soybean plants cannot with- stand the sweeping action of the net. Use other sampling methods for young plants.
- Sweep nets are difficult to use when plants are wet.
- Insects such as stink bugs, threecornered alfalfa hoppers, grasshoppers, and adult beetles can fly or move away from the sweep net before they are counted. (Insects are cold-blooded and become more active as temperatures increase during hot summer days.)
Ground Cloth Sampling Method
A ground cloth is an off-white fabric (usually cotton), 3 feet long on each side, with a ½- to ¾-inch dowel attached to each side. The dowels serve as handles to position the ground cloth on the soil.
For ground cloth sampling:

- Place the ground cloth flat on the soil between two rows ( 5).
- Vigorously shake the plants in 3 feet of row on both sides of the cloth
- Count the insects that fall onto the cloth
- Include any insects that fall at the base of plants. This gives the number of insects per 6 feet of row (3 feet of row on each side of the cloth).
- Take samples from multiple locations in the field. Unlike sweep net samples, these samples will consist of insects collected from the bottom to the top of the soybean plants.
- When sampling and kneeling on the ground, avoid fire ant mounds and possible stinging attacks if you disturb the mounds.
Advantages:
- Ground cloths are easy to use, inexpensive, and easy to carry into and out of the field.
Disadvantages:
- Ground cloths cannot be used in drill-planted soybeans unless the drill rows are widely spaced.
- Ground cloths cannot be used in wet fields.
- Each sample represents a relatively small sample area.
- Dislodged insects can fly or move away before being counted.
Maturity Groups, Determinate and Indeterminate Soybeans, and Growth Stages
Soybean characteristics and growth patterns relative to the plant- ing date can have significant impacts on pest populations. Frequent scouting for pests that attack during vulnerable soybean growth stages is critical in order to detect and control these pests when densities exceed treatment thresholds.
For instance, on the Upper Gulf Coast east of Houston, velvetbean caterpillars, green cloverworms, and soybean loopers often occur in August and September on soybean varieties planted in May and June that require a long time to mature.
However, varieties planted in April that have a shorter maturation period are not attractive to these caterpillar pests in August and September and often avoid this damage. But, stink bugs are most abundant early in the season and can be very damaging to these earlier maturing soybeans.
Soybeans develop vegetatively first, then reproductively. Flowering occurs during the reproductive growth stage. Day length, variety, and planting date all influence this sequential growth pattern. The soybean is a short-day plant; flowering is triggered when day length is less than a critical value. This means that soybeans adapted to the northern US require a longer minimum day length to initiate flowering compared to soybeans adapted to the southern US. Northern soybeans begin flowering later in the summer than do southern soybeans.
Soybeans are classified by maturity group (MG 00 through X). Smaller numbers or Roman numerals indicate varieties adapted to more northern latitudes and larger numbers or Roman numerals indicate varieties adapted to more southern latitudes. For instance, for the eastern half of the state, most Texas soybean farmers plant MG IV–VII varieties. When planted at the proper times, MG VI and VII varieties mature later than MG IV and V varieties.
Soybeans are also classified as determinate or indeterminate (Fig. 6). Determinate soybeans have a more defined period of vegetative and reproductive growth. Once flowering begins, little additional vegetative growth occurs. After initial flowering, indeterminate soybeans continue to produce flowers and foliage. Pods on indeterminate soybeans are more evenly distributed on the stem than those on determinate soybeans, which tend to clump at the top of the plant. Most early MG soybeans are indeterminate and most late MG soybeans are determinate.
When soybeans emerge from the soil, it is easy to see the cotyledons (paired, kidney-shaped food storage organs). This is the VE stage (Fig. 7). V stands for “vegetative.”


| Table 1. Soybean growth stages and accepted codes | |
| Fehr and Caviness (1997) | Plant Development |
| VE | Emergence |
| VC | Cotyledon + unfolding unifoliate leaves |
| V1 | First node trifoliate leaves + photosynthesis |
| V2 | Second node |
| V3 | Third node |
| V4 | Fourth node |
| V5 | Fifth node |
| V6 | Sixth node |
| V(n) | Nth node |
| R1 | Beginning bloom |
| R2 | Full bloom |
| R3 | Beginning pod development |
| R4 | Full pod |
| R5 | Beginning seed |
| R6 | Full seed |
| R7 | Beginning maturity |
| R8 | Full maturity leading to harvest |
| Fehr, W.R. and C.E. Caviness. 1977. Stages of Soybean Development. SR80, Iowa State University. The most modern refinement of this system is described in Ritchie, S. W., J. J. Hanway, H. E. Thompson, and G. O. Benson. 1989. How a Soybean Plant Develops. SR53, Iowa State University. | |
Then, in the VC stage, the first leaves to develop are unifoliate (a single, entire leaf), located on opposite sides of the young stem. Leaves in the V1 stage are trifoliate (consists of three leaflets). As the plant grows, additional trifoliate leaves grow at each node. The leaves alternate (adjacent nodes produce leaves originating from opposite sides of the stem) up the stem of the plant. Successive growth stages are identified by increasing numbers of trifoliate leaves (for example, V2- and V3-stage plants have two and three unrolled trifoliate leaves, respectively).
Once the plant begins flowering, plant growth stages are designated R1–8 (Table 1). R stands for “reproductive.” A growth stage begins when 50 percent or more of the plants are in or beyond that stage.
The time interval from planting to R1 and among the R stages can have a major influence on insect damage. The longer the plant is vulnerable to a particular pest, the greater the chance for damage. For instance, R3–6 growth stages are very susceptible to stink bug injury. Since this developmental period can last up to 7 weeks, regular and frequent scouting is essential.
